Returns to Patient Safety Evidenced through Infusion Data
by Michelle Santos, BScN, RN
Clinical Informatics Analyst, Infusion Devices & Data Management, Baxter Healthcare Corporation
Tim Hoh, BScPhm
Sr. Manager Pharmacy, Medical Affairs, Global Fluid Systems, Baxter Healthcare Corporation
John Soliven-Llaguno, BHScN, RN, NMD, PhD
Franchise Leader, Infusion Device & Data Management, Baxter Healthcare Corporation
Abstract
Assessment of infusion data from smart pumps offers evidence of underreported near miss events that are common within both the tertiary and community hospitals included in this study. The data demonstrates that a programming error occurs in 6% of infusions and is slightly higher in community hospitals compared to tertiary hospitals. The dose error reduction software (DERS) system alerts encouraged clinicians to reassess the programming and prompted a change in the majority of infusions (75%) demonstrating DERS alerts effectiveness and the smart pumps contribution to a safe culture. Evidence of averted harm is demonstrated through assessment of the drugs most commonly associated with near miss events as they are revealed to be amongst the top drugs associated most commonly with harm as reported medication errors to ISMP (2006).
The near miss events averted potential costs of approximately $800,000 (127 near miss events x $8750 per adverse event according to Hertzel & Sousa, 2009), associated with adverse medication errors as the unintended incorrectly programmed doses did not bring harm to the patient as the DERS system prompted the clinician to reassess and adjust the programming in the majority of cases. The data provides evidence to support a return on patient safety related to averting errors and promoting a safe culture and health economics returns. The future opportunity is for hospitals to learn from each other through benchmarking and transference of best practices amongst hospitals with similar challenges and profiles.
Keywords:
Dose Error Reduction Software (DERS), Smart IV Pump technology, Medication Error, Near Miss, Averted Medication Error, Health Economic Return
Background/Rationale
The 1999 Institute of Medicine (IOM) report provided groundbreaking statistical information related to adverse events and their harm based on United States (US) data, while the recent Canadian Adverse Event Study (Baker et al, 2004) quantified the potential risk of harm within Canada. These studies defined and detailed adverse events that led to subsequent studies in an attempt to find solutions and process improvements to reduce errors in healthcare. A proposed improvement toward mitigating risk was identified by the Ontario Health Technology Association of Ontario (OHTAC, 2009) which led to direction for hospitals to implement smart pumps containing dose error reduction software (DERS). While smart pump adoption has been identified as a first step towards averting medication errors, assessment of the infusion data provides additional patient safety returns through evidence of averted incidents. This article will explore infusion data specific to near miss events and the demonstrated return on investment to patient safety through the use of smart pump technology.
The IOM defines patient safety as the “prevention of harm to patients” with a focus on “the system of care delivery that (1) prevents errors; (2) learns from the errors that do occur and (3) is built on a culture of safety” (Mitchell, 2008, p. 1-2) Approximately two-thirds of preventable deaths were due to infusion therapy and most were attributable to manual programming errors when using infusion pumps (Healthcare Human Factors Group, 2009). Smart pump technology has been credited for mitigating risk of medication errors through programmable and customized safety limits for intravenous infusions that reduce potential human error (Murdoch & Cameron, 2008). Baxter’s uniquely Canadian Clinical Informatics program™ is dedicated to the analysis of infusion data obtained from Baxter infusion smart pumps containing DERS designed to prevent human error and avert potential harm. The program was created with the goal of improving patient safety and reducing harm through analysis of evidence-based infusion data in partnership with hospital safety initiatives. Current medication incident reporting systems are voluntary and frequently anecdotal; usually only reporting when an actual incident occurs that delivers harm to a patient (Louie, Wilmer, Wong, Grubisic, Najib & Dodek, 2010). By using infusion pump data, one is able to view evidence that may not be reported under the current voluntary reporting criteria because they are errors that were averted using DERS and did not deliver harm to the patient. However the rich data surrounding these near miss events can be critical to preventing future potential harm and demonstrating the efficiency of the software devices.
Method
Approximately 326,000 hours of infusion data was obtained from Baxter Colleague™ infusion devices at 30 hospital sites within Canada representing 19 tertiary and 11 community hospitals. Hospital clinical acuity and program offerings differ between tertiary and community hospitals, thus for the purposes of this clinical paper this classification distinction will be presented within the reported data to eliminate the possibility that any trends in data are related to this clinical distinction.
The data was processed based on event history within Baxter’s Colleague™ smart pumps using an algorithm database to classify key events, developed and assessed by Baxter’s Clinical Informatics program™. The program assesses a variety of alerts and clinician responses however for the purposes of this clinical paper data related to all infusions programmed within Baxter’s Colleague™ smart pump will be initially presented with a specific focus directed at the infusions programmed within the DERS system proprietarily named Guardian™. The infusion data provides evidence-based detail of medication errors that were averted; their potential associated harm, as well as the averted cost savings associated with using Colleague Guardian™ (DERS) smart pump software demonstrating a return on patient safety.
Method
Approximately 326,000 hours of infusion data was obtained from Baxter Colleague™ infusion devices at 30 hospital sites within Canada representing 19 tertiary and 11 community hospitals. Hospital clinical acuity and program offerings differ between tertiary and community hospitals, thus for the purposes of this clinical paper this classification distinction will be presented within the reported data to eliminate the possibility that any trends in data are related to this clinical distinction.
The data was processed based on event history within Baxter’s Colleague™ smart pumps using an algorithm database to classify key events, developed and assessed by Baxter’s Clinical Informatics program™. The program assesses a variety of alerts and clinician responses however for the purposes of this clinical paper data related to all infusions programmed within Baxter’s Colleague™ smart pump will be initially presented with a specific focus directed at the infusions programmed within the DERS system proprietarily named Guardian™. The infusion data provides evidence-based detail of medication errors that were averted; their potential associated harm, as well as the averted cost savings associated with using Colleague Guardian™ (DERS) smart pump software demonstrating a return on patient safety.
Definitions
The Colleague Guardian™ (DERS) software compares the dose a clinician programs with the hospitals pre-defined and customizable Guardian™ drug library limits. Creation of the drug library parameters requires a multi disciplinary team that incorporates clinical (nursing, physician and pharmacy) input to arrive at agreed-upon safe infusion limits for each identified care area. If any infusion is programmed outside of the dosing limits (defined by the clinical team within the drug library) the Guardian™ software produces an alert informing the clinician that the programmed dose is outside the predefined limit (see Illustration 1). At this stage the clinician is offered two options: to reprogram the dose to be within the predefined limits (considered a near miss) or to accept the dose outside of the predefined limits (considered an override). The Colleague™ pump can also be programmed to infuse within two other infusion modes: rate volume start which permits a clinician to program an infusion by entering the rate and volume of the infusion; and dose mode permits a clinician to enter a dose to infuse which is calculated by the pumps internal software once the clinician enters the amount of drug within the diluent volume of the intravenous (IV) bag. Optional modes of infusion ensure the availability to infuse a medication that may not be listed within the hospitals predefined drug library or formulary, such as study drugs or fluids lacking medications (for hydration etc.)
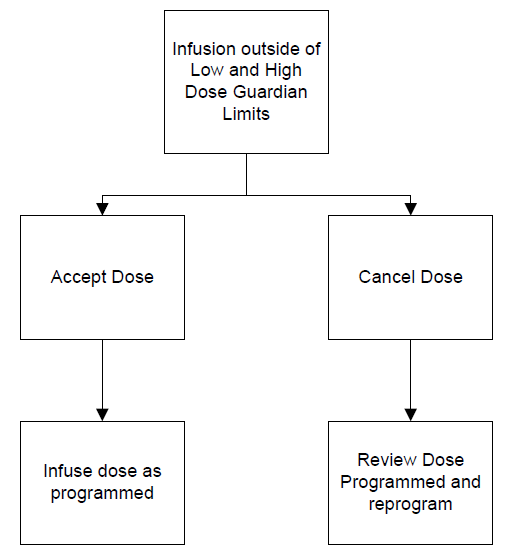
Illustration 1: Guardian™ software decision tree
Results
Analysis of the modes of infusion selected for programming is classified by tertiary versus community hospital within Figure 1.
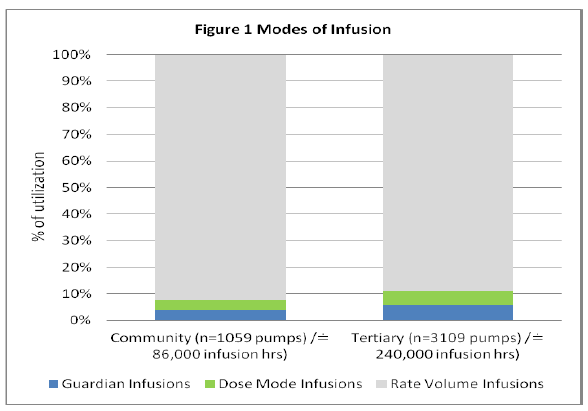
Figure 1: Modes of Infusion
The majority of infusions were programmed within the Rate Volume mode which has no DERS capability and this programming pattern is consistent between community and tertiary sites. Guardian™ utilization appears low when compared to rate volume start modes and although not within the scope of this paper this could be investigated in future. Possible explanations could include a predominance of infusing basic intravenous fluids that do not contain drugs. Figure 2 depicts the percentage of infusions that were programmed using the Guardian™ DERS system (Tertiary 6%, Community 4%).
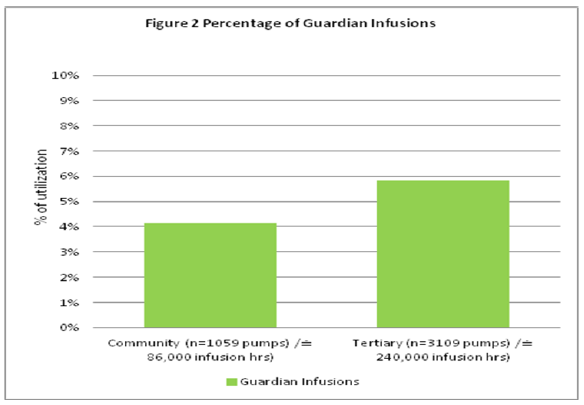
Figure 2: Percentage of Guardian Infusions
Detailed analysis of these near miss events within the Guardian™ DERS programming provides insight related to returns on patient safety and will be the focus of this clinical paper. Figure 3 depicts the percentage of Guardian™ infusions associated with a near miss event (Tertiary 6%, Community 7%).
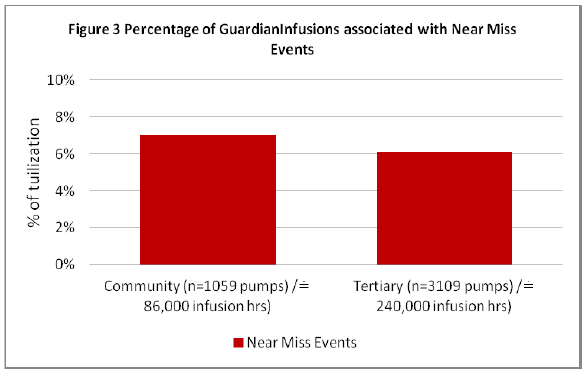
Figure 3: Percentage of Guardian Infusions associated with Near Miss Events
The data demonstrates similar results between community and tertiary hospitals near miss events, however it is noted that although the tertiary sites had increased Guardian™ utilization (Figure 2) it is the community hospital which demonstrated increased near miss events (Figure 3) when programming within Guardian™. Combining the data for all sites demonstrates of all infusions programmed within the Guardian™ DERS systems there is a 6% incidence of near miss event associated with programing an infusion (Figure 9).
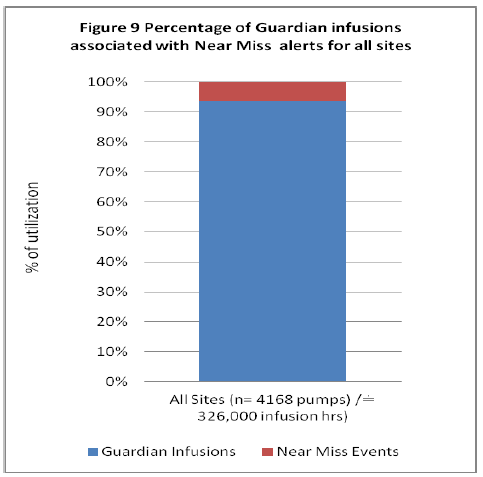
Figure 9: Percentage of Guardian Infusions associated with Near Miss Events for all sites
Further analysis regarding all near miss events and actions during programing elicits detail related to actions the alerts prompted, in addition to any harm associated with the initial programming that prompted the DERS alert.
Near Misses
Of the total 127 near miss alerts the majority (66) were initially programmed above the high dose limit within the Guardian™ DERS software and on average they were programmed at doses 8 times above the intended dose (based on initial programmed dose compared to Guardian™ drug library high dose limit). The remaining 61 infusions were initially programmed at doses below the drug library low dose limit, and on average the doses were 16 times below the Guardian™ low dose limit (based on initial programmed dose compared to Guardian™ drug library low dose limit) (Refer to Figure 4).
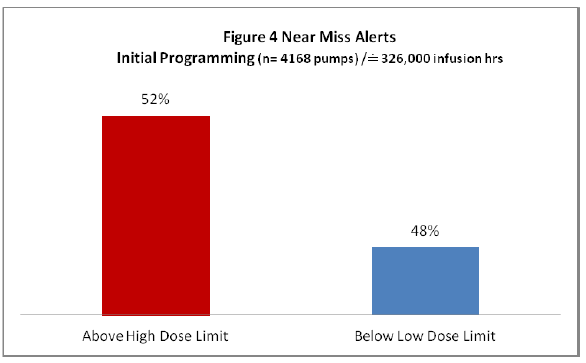
Figure 4: Near Miss Alerts
Once the clinician selected the option to reassess the infusion dose parameters Figure 5 depicts the actions taken (“other” represents situations where the pump was turned off or the infusion was cancelled).
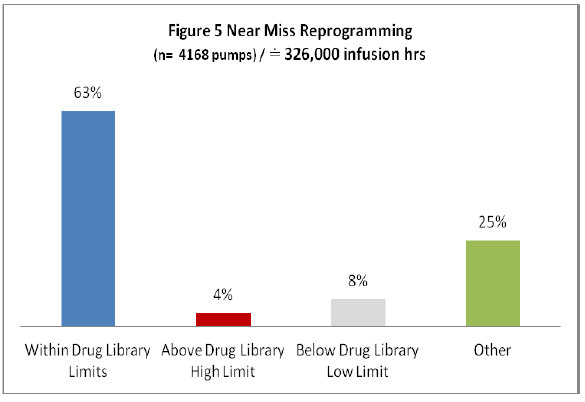
Figure 5: Near Miss Reprogramming
Of all near miss alerts the majority (75%) were reprogrammed indicating that the initial programming was erroneously entered and the Guardian™ alert obligated the clinician to reassess and reprogram the infusion. While there were some infusions that were reprogrammed outside of the hospital defined limits (which may have been deemed clinically appropriate) the majority (63%) were programmed within. Examples of averted programming errors (Figure 6) identify initial programming compared to final infusion dose representing evidence of averted overdose and under dose scenarios (when compared to doses deemed appropriate by the hospitals defined dose parameters within their Guardian™ drug library).
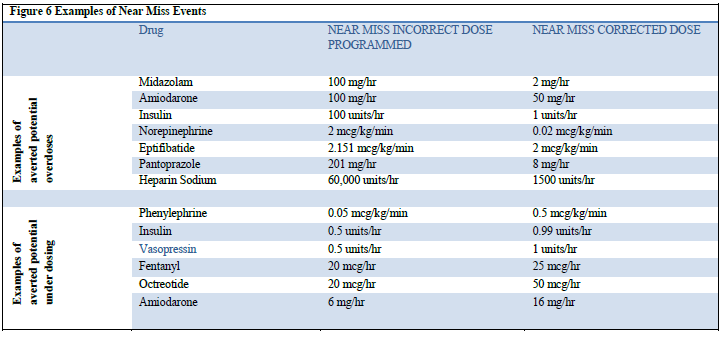
Figure 6: Examples of Near Miss Events
Analysis regarding the drugs associated with the near miss event programming provides detail regarding harm. Figure 7 depicts the top drugs associated with a near miss event.
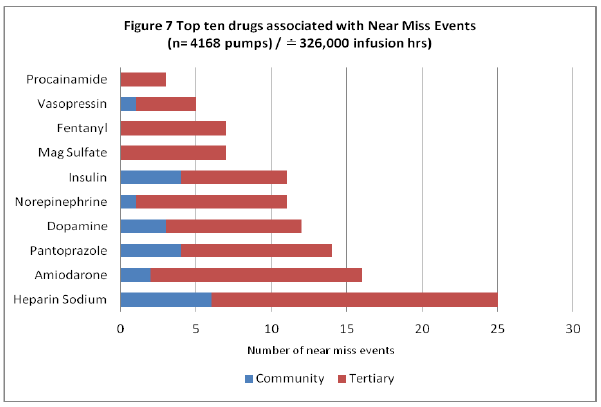
Figure 7: Top Ten Drugs Associated with Near Miss Events
The majority of drugs associated with a near miss event within the infusion data (Figure 7) are also identified as ISMP high alert medications, specifying the need for vigilant care when running such medications as incorrect dosing can lead to significant harm (ISMP, 2008). The infusion evidence demonstrates a correlation between the top ten drugs associated with a near miss event and the top ten ISMP commonly reported drugs associated with harm (through voluntary reporting of medication errors) (ISMP, 2006).
The most common drug associated with a near miss event within the data is Heparin Sodium, identified by ISMP as one of the top ten frequently reported medications causing harm when delivered erroneously (ISMP, 2006). The detail of Heparin Sodium near miss events and their reprogramming are depicted in Figure 8.
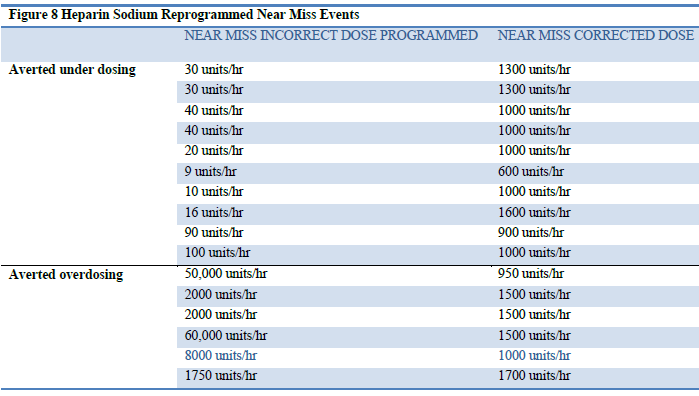
Figure 8: Heparin Sodium Reprogrammed Near Miss Events
Understanding the common errors that occur while programming an infusion provides a first step towards prevention of future inadvertent errors. Murdoch and Cameron (2008) identified the three most common types of pump errors as multiple of ten errors, unit errors, and push button errors. Multiple of ten errors often occur when an infusion is set up with a dose or time to infuse that is a multiple of ten higher or lower than the correct prescription. The second type of error, unit errors, occurs when there is a mistake involving units of dose, weight or time. The final type of error is a push button error which occurs when a clinician pushed a wrong button when programming an infusion.
Within the Heparin infusions there was evidence of each of the common errors identified by the literature, however there were no errors related to selecting the wrong dose mode (unit) because the drug library acts as a safety net within the Guardian™ DERS system associating the pharmacy defined dose mode (unit) for each drug to prevent this error. This feature combined with the customizable dose limits ensure infusions are delivered within the hospital defined safe limits regardless of the common programming error that may be made by a clinician. Although not within the scope of this clinical paper the opportunity for research into near miss common programming errors using infusion data provides insight beyond currently reported anecdotal accounts ensuring healthcare is “learning from errors that do occur” (Mitchell, 2008, p. 1-2) as well as from those that are averted.
Analysis of infusion data provides the impetus to begin to understand the near miss events which are replete with rich data that can be applied to develop improved future enhancements to current safety systems.
Discussion
Information related to medication errors within Canada obtained from studies such as the Canadian Adverse Event Study (Baker & Norton, 2004) and through ISMP’s encouraged voluntary reporting of medication errors (ISMP, 2010) identifies medication errors, which have delivered harm. These reported errors provided the impetus for healthcare to develop safety systems that incorporated technology such as smart pumps to improve the safety of infusion medication delivery. While averting harm through identified errors is beneficial to future avoidance, it is the assessment of evidence based infusion data events specific to near misses that demonstrate an underreported return on patient safety. Analysis of Baxter’s Colleague™ DERS infusion data representative of 30 Canadian hospital sites demonstrates a return on patient safety through averted harm to patients and its associated financial implications of said harm.
Within the data gathered from all infusions programmed within DERS systems there was a demonstrated 6% incidence of near miss events (Figure 9): of these events, the majority (63%) were reprogrammed within the hospital defined drug library safe doses supporting a return on patient safety through averted errors. Interestingly, there was evidence of higher incidents of near miss events within the community sites (when compared to the tertiary sites) even though they had lower Guardian™ DERS use – although this is not within the scope of this clinical paper, future research to identify reasons for this could be investigated. Patient safety as defined by the IOM includes a commitment to a “culture of safety.” Any hospital’s demonstrated practice of programming drugs within DERS systems combined with infusion data analysis justifies its use, and is further enhanced by the evidence for averted unintended programming errors (Mitchell, 2008, p. 1-2).
The majority of the top ten drugs associated with near miss events are also high alert medications according to ISMP (2008) indicating a need for vigilance when programming as these drugs can elicit significant harm if provided erroneously. Specific drugs identified within the data: (Figure 7) Heparin, Insulin and Fentanyl are known by ISMP (2006) to have higher reported patient harm when associated with a medication error. The data also identified these drugs to be amongst the top drugs associated with a near miss event, therefore demonstrating harm avoidance through use of DERS systems (ISMP, 2006). Data analysis of the top drugs involved in a near miss event provides evidence of a hospital’s demonstrated commitment to establishing a “culture of safety” by ensuring these drugs have defined safety dosing parameters within their drug libraries and by encouraging use by clinicians to keep within these dose ranges (Mitchell, 2008, p. 1-2).
Bullock (2011) identified barriers to voluntary reporting systems, such as clinician shame and fear associated with near miss events, and justified and reinforced the need for evidence-based near miss reporting structures to capture true data of actual events. Clinician reassessment of infusion alerts for programming mismatches with hospital identified drug library parameters offer the clinician the opportunity to safely review a potential error and perform the necessary correction. This near miss may not be considered reportable as Bullock (2011) identified clinicians’ confusion with reporting parameters surrounding near miss events. With the knowledge of smart pump infusion data analysis the clinician and hospital can be comforted to know that the alert was avoided and the data captured within the device can be appropriately assessed at a later time. The IOM relates patient safety to learning from “errors that do occur” however the opportunity to learn from near miss events through infusion data demonstrates a return on patient safety that did not bring harm to the patient and promotes a continued “culture of safety” (Mitchell, 2008).
Heparin is associated with the highest incidence of near miss events in both tertiary and community hospital sites. The detail surrounding the common programming errors provides rich data as an impetus towards investigating safety system improvements specific to its programming that can be investigated further in other studies.
Adverse events generate financial burden through extended hospital stays linked to additional treatments and investigations. Hertzel and Sousa (2009) monetized adverse events costs at an estimated $8750 (per event). Using this financial estimate the Colleague™ data demonstrated approximately $830,000 (reprogrammed 95 near miss alerts x $8750) in averted medication error expenditures. The return on patient safety from a health economics perspective is evidenced through averting harm and its associated financial implications meeting the patient safety objective of preventing “harm to patients” (Mitchell, 2008, p. 1-2).
Conclusion
Reviewing infusion data within hospitals provides evidence of near miss events that avert harm and its associated costs within both community and tertiary institutions (as the data demonstrates similar results within both hospital categories).
The dissemination of hospital infusion data results provides a mechanism for clinicians to learn from other’s near miss errors demonstrating an additional return on patient safety opportunity in avoiding common pitfalls and potentially dangerous mistakes.
While the data provides evidence to support a return on patient safety related to averting errors, promotes a safe culture and a health economic return, future opportunities exist for hospitals to learn from each other through benchmarking and transference of best practices from hospitals with similar challenges and profiles.
References
Baker et al, (2004). The Canadian Adverse Events Study: the incidence of adverse events among hospital patients in Canada. Canadian Medical Association Journal, 170 (11), 1678-1686.
Bullock, L.M., (2011). Transform into a culture of safety. Nursing Management, 42 (7), 14-16.
Healthcare Human Factors Group, (2009). Smart medication Delivery Systems: Infusion Pumps . Toronto: University Health Network. Retrieved from http://www.ehealthinnovation.org/files/SmartMedicationDeliverySystems_FullReport.pdf
Hertzel, C., & Sousa, V., (2009). The Use of Smart Pumps for Preventing Medication Errors. The Journal of Infusion Nursing. 2009; 32, (5). 257-267
Institute for Safe Medication Practices (2008). ISMP’s List of High-Alert Medications http://www.ismp.org/tools/highalertmedications.pdf
Institute for Safe Medication Practices Safety Bulletin (2006). Top 10 drugs reported as causing harm through medication error. Vol 6, Issue 1. http://www.ismp-canada.org/download/safetyBulletins/ISMPCSB2006-01Top10.pdf
Institute for Safe Medication Practice (2007). Medication Safety Alert: Smart pumps are not smart on their own. Retrieved http://www.ismp.org/newsletters/acutecare/articles/20070419.asp
Institute for Safe Medication Practices (2009). Medication Errors and Risk Management in Hospitals. http://www.ismp-canada.org/Riskmgm.htm
Institute for Safe Medication Practices (2010). Medication Incident and Near Miss Reporting Program. Retrieved from https://www.ismp-canada.org/err_ipr.htm
Institute of Medicine (1999). To Err is Human http://iom.edu/~/media/Files/Report%20Files/1999/To-Err-is-Human/To%20Err%20is%20Human%201999%20%20report%20brief.pdf
Louie, K., Wilmer, A., Wong, H., Grubisic, M., Najib, A. & Dodek, P. (2010). Medication Error Reporting Systems: A survey of Canadian Intensive Care Units http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2832561/
MacPhee, E., Sherrard, H., (2007). An Innovative Approach to ‘Near Miss’ capture for improvement of patient safety. Retrieved from Canadian Patient Safety Institute http://www.patientsafetyinstitute.ca/english/search/pages/default.aspx?k=macphee
Mitchell, P., (2008). Patient Safety and Quality: An evidence based handbook for nurses. Retrieved from http://www.ncbi.nlm.nih.gov/books/NBK2681/
Murdoch, L., Cameron, V., (2008). Smart Infusion Technology: A minimum safety standard for intensive care. British Journal of Nursing, Vol. 17, Iss. 10, 22 May 2008, pp 630 – 636
Ontario Health Technology Advisory Board, (2009). Recommendation: Implementation and Use of Smart Medication Delivery Systems. Retrieved from http://www.health.gov.on.ca/english/providers/program/ohtac/tech/recommend/rec_smd_20090701.pdf
Author Bios
Michelle Santos, BScN, RN
Michelle is a Bachelors prepared Registered Nurse with a specialty in critical care from Humber College. Her critical care clinical experience includes Neurology, Cardiovascular and currently practicing within a Medical Surgical Transplant critical care unit. She has developed national experience in informatics and continues to develop innovations within Baxter’s data management programs while providing both clinical and analytics consulting within Baxter Healthcare Corporation. She continues to develop her expertise and is currently working towards obtaining her Masters of Nursing.
Tim Hoh, BScPhm
Tim is a Pharmacist with over 20 years experience in Clinical and Pharmaceutical Industry settings. He possesses thorough knowledge of risk assessments and hazard analysis of medical systems and infusion devices. Tim’s expertise include best practices for Continuous Quality Reporting, Drug library development for infusion devices, pharmacy aseptic compounding, USP797 guidelines and pharmacy cleanroom designs.
John Soliven-Llaguno, BHScN, RN, NMD, PhD
John is a Bachelors prepared Registered Nurse with a specialty in critical care from the British Columbia Institute of Technology, with post graduate studies in experimental, osteopathic & naturopathic medicine. He has worked in health care for over twenty years including positions of escalating responsibilities in medical device and pharmaceutical companies and most recently at Baxter Health Care Corporation. He is the primary architect for the development of Clinical Informatics and has a passion for primary care medicine.







